Low-dose endoxifen breast density reduction is emerging as a promising development in breast cancer prevention research, following a new clinical study suggesting it may match the effectiveness of tamoxifen while producing fewer side effects.
A recent study conducted by researchers at Karolinska Institutet has foundendoxife that low doses of endoxifen can reduce breast density to a similar level as the widely used drug tamoxifen, but with a potentially improved side effect profile. The findings were published in the Journal of the National Cancer Institute and may influence future preventive treatment strategies for breast cancer.
Understanding the Study on Breast Density and Treatment
The research focused on how low-dose endoxifen breast density reduction compares with standard tamoxifen therapy. Breast density is a key factor in breast cancer risk, as higher density is associated with increased likelihood of developing the disease.
Why Breast Density Matters
Breast density refers to the proportion of fibroglandular tissue compared to fatty tissue in the breast. Higher density can:
- Make tumors harder to detect on mammograms
- Increase the risk of developing breast cancer
- Serve as a measurable indicator in prevention studies
Reducing breast density is therefore considered an important goal in preventive oncology research.
Investigational Role of Endoxifen
Endoxifen is the most active metabolite produced when tamoxifen is broken down in the body. Researchers have been investigating whether administering endoxifen directly could provide more predictable results and improved tolerability.
In this low-dose endoxifen breast density study, scientists aimed to determine whether small doses could achieve similar biological effects without the intensity of tamoxifen-related side effects.
How the Clinical Trial Was Conducted
The study was designed as a proof-of-concept trial, meaning it aimed to confirm biological effectiveness before larger clinical trials are conducted.
Study Design and Participants
A total of 240 healthy premenopausal women participated in the trial. They were randomly assigned into three groups:
- Placebo group
- 1 mg endoxifen daily group
- 2 mg endoxifen daily group
The treatment period lasted six months, during which researchers closely monitored changes in breast density and reported side effects.
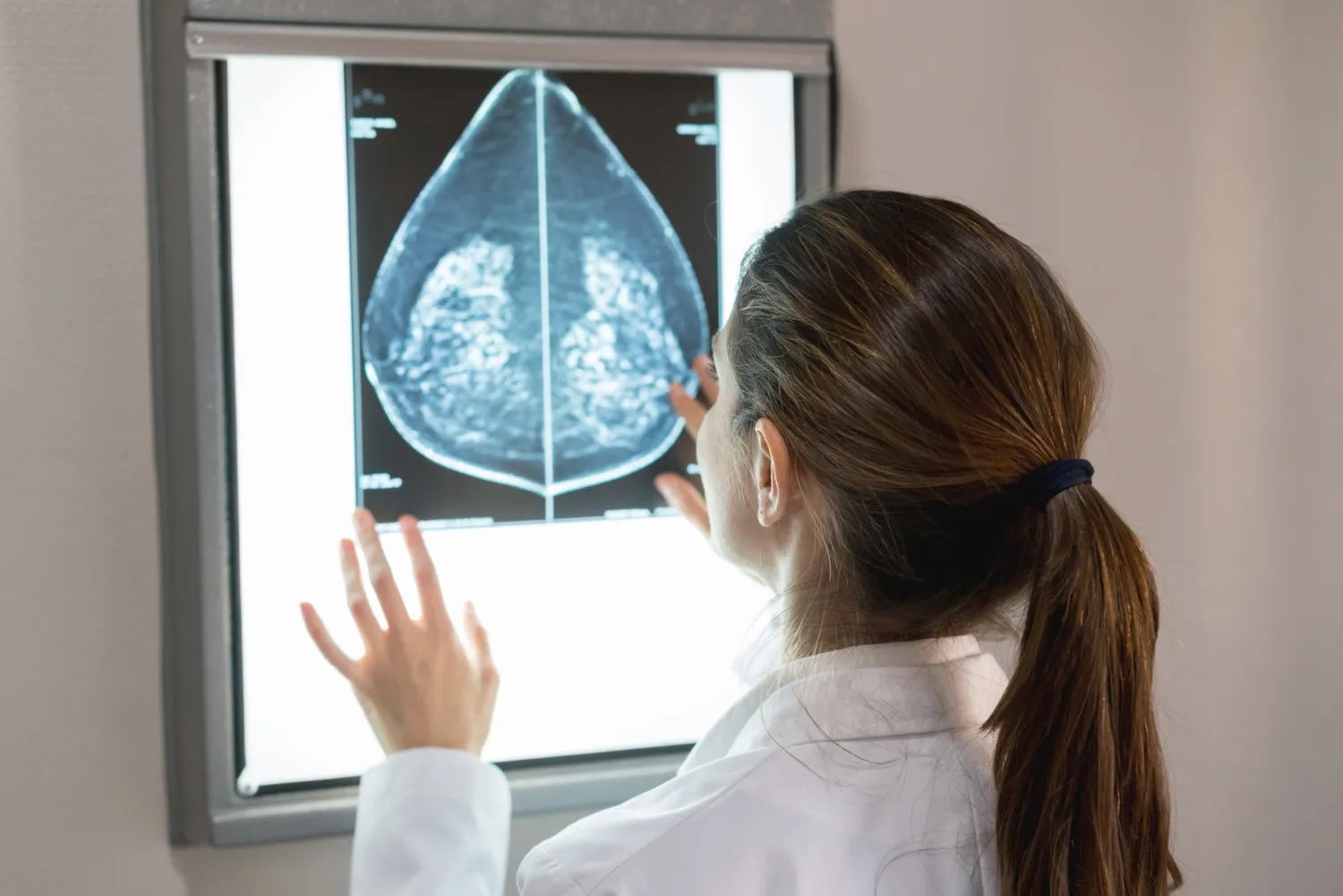
Measurement Approach
Researchers used mammographic imaging to assess changes in breast density. This allowed them to evaluate how effectively low-dose endoxifen breast density reduction compared to both placebo and known tamoxifen results from previous studies.
Key Findings From the Research
The results showed a clear and measurable reduction in breast density among participants receiving endoxifen.
Breast Density Reduction Results
The study reported the following outcomes:
- 1 mg endoxifen: approximately 19% reduction in breast density
- 2 mg endoxifen: approximately 26% reduction in breast density
- Placebo group: no significant change
For comparison, previous research shows that 20 mg of tamoxifen reduces breast density by around 18.5%. This suggests that low-dose endoxifen breast density reduction may be at least as effective as standard tamoxifen dosing.
Comparison With Tamoxifen
The findings indicate that:
- Both low-dose endoxifen groups achieved similar or stronger results than tamoxifen
- Even minimal dosing produced measurable biological effects
- The response appeared consistent across participants
These results are important because they suggest a potentially more efficient therapeutic approach.
Side Effects and Safety Profile
One of the major concerns with tamoxifen is its side effect burden, which often leads to discontinuation of treatment.
Common Tamoxifen Side Effects
Tamoxifen is known to cause:
- Hot flushes
- Night sweats
- Menopause-like symptoms
- General discomfort leading to reduced adherence
These side effects have limited long-term preventive use for some patients.
Endoxifen Tolerance in the Study
The study showed a difference in tolerability between doses:
- The 1 mg group reported side effects similar to placebo
- The 2 mg group experienced more hot flushes and night sweats
- Serious adverse effects were not significantly different across groups
This suggests that low-dose endoxifen breast density reduction may be achievable with a more favorable safety profile, especially at lower doses.
What These Findings Could Mean for Breast Cancer Prevention
The study highlights the potential for endoxifen to become part of future preventive strategies for breast cancer.
Possible Clinical Advantages
If confirmed in larger trials, endoxifen could offer:
- Comparable effectiveness to tamoxifen
- Improved patient tolerance at lower doses
- More predictable biological response
- Better long-term adherence rates
These factors are critical in preventive treatments, where patients are typically healthy but at elevated risk.
Importance of Treatment Completion
One of the challenges with tamoxifen is that many patients discontinue use due to side effects. A better-tolerated option could improve completion rates and overall prevention outcomes.
Low-dose endoxifen breast density reduction could therefore represent a meaningful shift in how risk reduction therapies are delivered.
Study Limitations and Future Research
While the findings are promising, researchers emphasize that this is an early-stage study.
Key Limitations
- The trial only lasted six months
- Participants were healthy premenopausal women
- Cancer outcomes were not measured
- Long-term safety remains unknown
What Comes Next
Further research will be needed to determine:
- Whether endoxifen reduces actual breast cancer risk
- Long-term safety of low-dose use
- Optimal dosing strategies
- Effectiveness in high-risk populations
Large-scale clinical trials will be necessary before the treatment can be widely recommended.
Frequently Asked Questions (FAQ)
What is low-dose endoxifen breast density reduction?
It refers to the use of small doses of endoxifen to reduce breast density, which is linked to breast cancer risk.
How does endoxifen compare to tamoxifen?
Studies show that low-dose endoxifen can reduce breast density at a similar level to tamoxifen but may cause fewer side effects at lower doses.
Can endoxifen prevent breast cancer?
Current research shows promise, but it has not yet been proven to reduce breast cancer risk directly.
Why is breast density important in cancer prevention?
Higher breast density is associated with increased cancer risk and can make tumors harder to detect early.
Conclusion
The findings on low-dose endoxifen breast density reduction suggest a potentially important development in preventive breast health strategies. While tamoxifen remains a well-established treatment, endoxifen may offer similar benefits with improved tolerability at lower doses. However, further long-term studies are needed before conclusions can be made about its role in reducing breast cancer risk.
Click here for more news